Chelating ion exchange resin represents a crucial advancement in separation science, offering a highly selective and efficient method for removing specific metal ions from solutions. Its applications span diverse industries, from water purification and hydrometallurgy to biomedical applications and environmental remediation. Understanding its principles and capabilities is paramount for addressing global challenges related to resource management, pollution control, and human health.
The increasing demand for sustainable solutions and the growing scarcity of critical metals are driving the adoption of chelating ion exchange resin technologies. Traditional methods often lack the specificity and efficiency required for modern applications, leading to environmental concerns and economic inefficiencies. This resin provides a targeted approach, minimizing waste and maximizing resource recovery, aligning with principles of a circular economy.
Furthermore, the continuous innovation in resin design and manufacturing processes is expanding the scope of its applicability. New materials and functionalization techniques are enabling the development of resins tailored to specific separation needs, enhancing performance and reducing costs. This makes chelating ion exchange resin a cornerstone technology for a wide range of industries seeking sustainable and cost-effective solutions.


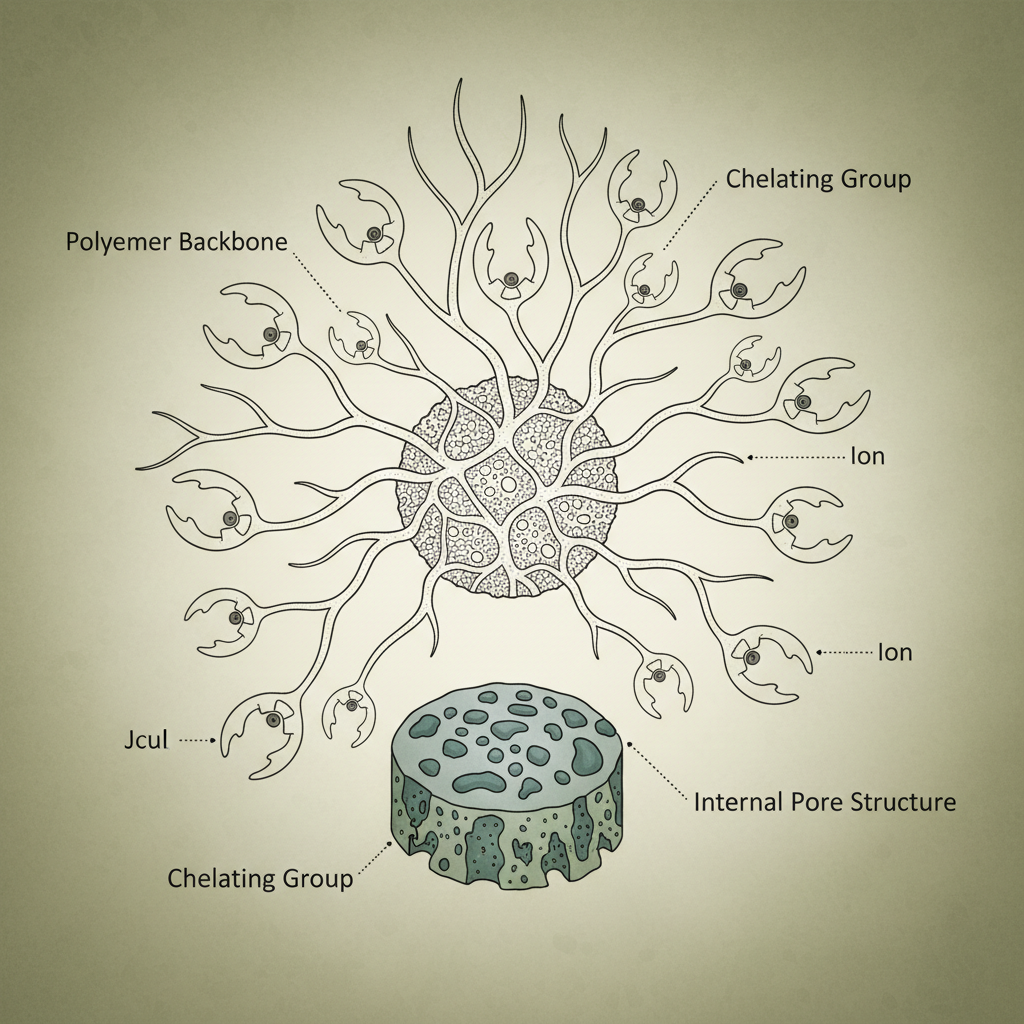
Chelating ion exchange resins are uniquely engineered materials designed to selectively bind and remove specific metal ions from liquid streams. Unlike traditional ion exchange resins that rely on electrostatic attraction, these resins incorporate chelating functional groups – molecules that form stable, ring-like complexes with target ions. This offers a significantly higher degree of selectivity, crucial for applications where precise separation is required.
The development of chelating ion exchange resin technology has been driven by the growing need for efficient and environmentally responsible methods for metal recovery, purification, and waste treatment. It’s a key component in tackling challenges across diverse fields, contributing to a more sustainable and resource-efficient future.
Essentially, a chelating ion exchange resin is a polymeric material—often a crosslinked polystyrene matrix—to which chelating agents are chemically bonded. These chelating agents, such as iminodiacetic acid (IDA), nitrilotriacetic acid (NTA), or ethylenediaminetetraacetic acid (EDTA), possess multiple donor atoms that can simultaneously coordinate with a metal ion. This forms a stable chelate complex, effectively capturing the ion from solution.
This process is integral to modern industry and critical for addressing humanitarian needs. In water treatment, it removes toxic heavy metals. In mining, it aids in the selective extraction of valuable metals. In medicine, it's used for detoxification and diagnostic purposes. Its versatile nature stems from the ability to tailor the chelating agent to target specific ions with exceptional accuracy.
The functionalization of the resin matrix with these chelating agents distinguishes it from conventional ion exchange resins, enhancing its selectivity and making it indispensable for complex separation processes.
Several key factors determine the effectiveness and suitability of a chelating ion exchange resin for a given application. These include the type of chelating agent, the resin matrix, the pore size distribution, and the mechanical stability of the beads. The choice of chelating agent dictates the selectivity for specific metal ions, while the resin matrix provides the structural support and influences the resin's physical properties.
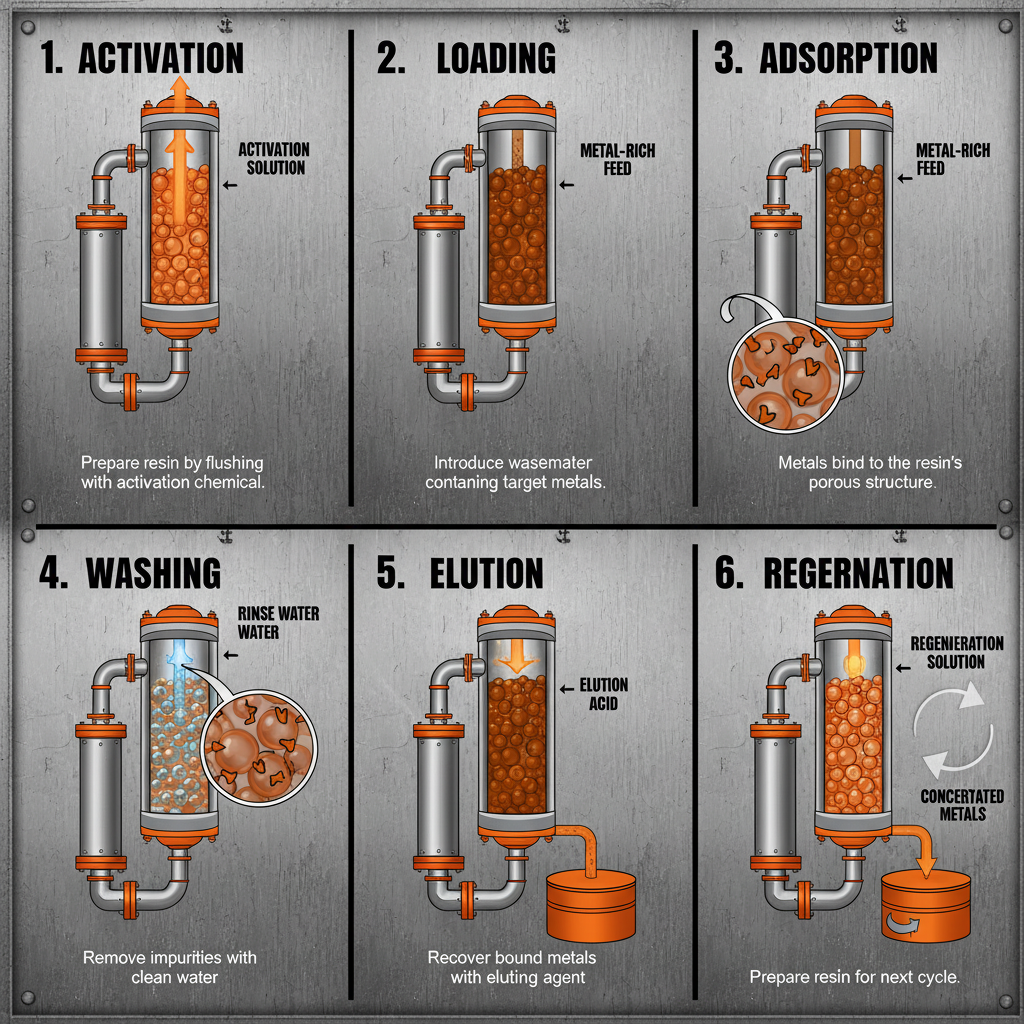
Durability is paramount; the resin must withstand repeated cycles of adsorption, elution, and cleaning without significant degradation. Scalability refers to the ease with which the resin can be produced in large quantities to meet industrial demands. Cost-efficiency is also critical, influencing the economic viability of the separation process. Finally, flow rate and pressure drop need to be optimized for efficient operation within a given system.
These components collectively contribute to the overall performance of the resin, enabling it to address complex separation challenges across a wide range of industries.
The applications of chelating ion exchange resin are remarkably diverse. In the hydrometallurgical industry, it's used for the selective recovery of precious metals like gold, platinum, and palladium from ores and recycled materials. In the nuclear industry, it plays a critical role in the treatment of radioactive waste, removing harmful isotopes from contaminated water.
Water purification represents a significant area of application, where these resins are used to remove heavy metals like lead, mercury, and cadmium, ensuring safe drinking water supplies. The pharmaceutical industry utilizes them for the purification of drug intermediates and the removal of metal contaminants. In post-disaster relief operations, portable systems employing chelating resins can provide rapid access to safe drinking water in affected areas.
Globally, countries with significant mining activities, such as Chile, Peru, and Australia, heavily rely on this technology for metal recovery. Regions facing water scarcity and pollution challenges, like parts of Africa and Asia, are increasingly adopting these resins for water treatment. Organizations such as the World Health Organization (WHO) promote the use of effective water purification technologies, highlighting the importance of chelating ion exchange resin in addressing global health concerns.
The benefits of using chelating ion exchange resin extend beyond just technical efficiency. From a cost perspective, the ability to selectively recover valuable metals can significantly reduce operational expenses and generate revenue. The minimized waste production translates to lower disposal costs and enhanced environmental compliance.
Sustainability is a major advantage. By enabling the recovery and reuse of critical resources, these resins contribute to a circular economy and reduce reliance on virgin materials. From a social impact perspective, ensuring access to clean water and mitigating environmental pollution directly improves public health and well-being. Long-term reliability and minimal maintenance requirements further contribute to its value.
Ongoing research focuses on developing novel chelating agents with even greater selectivity and affinity for target ions. The integration of nanomaterials into the resin matrix is enhancing its surface area and adsorption capacity. Furthermore, the application of machine learning algorithms is enabling the optimization of resin design and process parameters for specific applications.
The move towards “smart” resins capable of responding to environmental stimuli, such as pH or temperature, is also gaining traction. These innovations align with broader trends in green chemistry and sustainable manufacturing, paving the way for more efficient and environmentally responsible separation technologies.
One key challenge lies in the potential for fouling – the accumulation of organic matter or other contaminants on the resin surface, reducing its efficiency. Pre-treatment of feed streams and regular cleaning protocols can mitigate this issue. Another challenge is the cost of certain chelating agents, which can impact the overall economics of the process. Research into more affordable and sustainable alternatives is ongoing.
The development of more robust and mechanically stable resins is also crucial for extending their lifespan and reducing the need for frequent replacements. Finally, optimizing the regeneration process – the removal of adsorbed ions from the resin – is essential for minimizing waste and maximizing resource recovery.
By addressing these challenges through continued innovation and process optimization, the full potential of chelating ion exchange resin can be realized.
| Challenge | Impact on Performance | Potential Solution | Implementation Cost (1-10) |
|---|---|---|---|
| Resin Fouling | Reduced Adsorption Capacity | Pre-treatment of Feed Stream | 4 |
| High Chelating Agent Cost | Increased Operational Expenses | Research into Alternatives | 7 |
| Mechanical Instability | Reduced Lifespan | Improved Resin Matrix Design | 6 |
| Inefficient Regeneration | Increased Waste Generation | Optimization of Elution Conditions | 5 |
| Selectivity Limitations | Cross-Contamination Issues | Development of Novel Chelating Agents | 8 |
| Scale-Up Challenges | Difficulty in Large-Scale Production | Process Intensification Techniques | 6 |
Chelating ion exchange resins can be tailored to remove a wide range of metal ions, including heavy metals like lead, mercury, cadmium, copper, and zinc. The selectivity depends on the chelating agent used. Resins incorporating IDA, for example, exhibit strong affinity for copper, nickel, and zinc, while those using DTA are more effective for lead and cadmium. The ability to specifically target certain ions makes them ideal for applications requiring high purity or regulatory compliance.
The pore size of the resin matrix is a critical factor, particularly when dealing with complex samples containing large molecules or colloidal particles. Smaller pore sizes offer higher selectivity but may limit access to the chelating sites for larger ions. Larger pore sizes allow for the adsorption of larger molecules but may reduce selectivity. Choosing the appropriate pore size is essential for optimizing both capacity and selectivity based on the target application.
The lifespan of a chelating ion exchange resin varies depending on several factors, including the feed stream composition, operating conditions (pH, temperature, flow rate), and regeneration frequency. With proper maintenance and regeneration, a resin can typically last for several years. However, degradation of the matrix and loss of chelating agent functionality can eventually necessitate replacement. Regular monitoring of performance indicators is crucial for determining when replacement is required.
Yes, most chelating ion exchange resins can be regenerated, allowing for multiple adsorption-desorption cycles. Regeneration typically involves eluting the adsorbed metal ions from the resin using a suitable chemical solution, such as an acid or a complexing agent. The choice of eluent depends on the metal ion and the chelating agent. Effective regeneration is crucial for minimizing waste and maximizing the economic viability of the process.
Chelating ion exchange resins exhibit significantly higher selectivity than traditional ion exchange resins. Traditional resins rely primarily on electrostatic interactions, which are less specific. Chelating resins, on the other hand, utilize the formation of stable chelate complexes, resulting in a much stronger and more selective binding of target metal ions. This makes them ideal for applications where separation of closely related metals is required.
Environmental considerations include the safe handling of regeneration eluents and the proper disposal of spent resins. Eluents often contain concentrated metal ions and should be treated appropriately to prevent environmental contamination. Spent resins may be classified as hazardous waste depending on the adsorbed metals and should be disposed of in accordance with local regulations. Sustainable practices, such as resin regeneration and metal recovery from spent resins, are encouraged to minimize environmental impact.
In conclusion, chelating ion exchange resin represents a powerful and versatile technology with far-reaching implications for a wide range of industries. Its unique ability to selectively remove and recover metal ions offers significant advantages over traditional separation methods, contributing to improved efficiency, sustainability, and environmental protection. From water purification and resource recovery to pharmaceutical manufacturing and environmental remediation, these resins play a critical role in addressing global challenges.
Looking ahead, continued innovation in resin design, coupled with advancements in process optimization and data analytics, will further enhance the performance and expand the applicability of this technology. By embracing these advancements and promoting sustainable practices, we can unlock the full potential of chelating ion exchange resin and create a more resource-efficient and environmentally responsible future. Visit our website at www.lijiresin.com to learn more.