Iminodiacetic acid resin (IDAD) represents a critical class of chelating resins with widespread applications in various industrial and environmental processes. Its ability to selectively bind metal ions makes it invaluable in purification, separation, and recovery operations. The demand for efficient and sustainable solutions for metal management is continually increasing globally, driven by stricter environmental regulations and the need for resource conservation. Understanding the properties and applications of IDAD resin is therefore crucial for engineers, chemists, and environmental scientists alike.
Globally, the market for ion exchange resins, which includes IDAD resin, is experiencing significant growth, estimated to reach $7.2 billion by 2028 (Grand View Research, 2021). This growth is fueled by increasing demand in water treatment, pharmaceutical production, food and beverage processing, and hydrometallurgy. Furthermore, the growing need to remove heavy metals from wastewater and industrial effluents is a major driver. The United Nations Sustainable Development Goal 6 (Clean Water and Sanitation) emphasizes the importance of sustainable water management, directly benefiting from technologies utilizing resins like iminodiacetic acid resin.
IDAD resin plays a pivotal role in addressing these challenges. It's not simply a chemical compound but a key component in developing advanced solutions for resource recovery and pollution control. The resin's versatility and efficiency make it a cornerstone of modern chemical engineering practices, impacting industries ranging from mining to healthcare, and contributing to a more sustainable future.

The history of iminodiacetic acid resin development is intertwined with the evolution of ion exchange technology. Initially, naturally occurring materials like zeolites were used for selective ion absorption. However, the limitations in their selectivity and stability drove the development of synthetic resins in the mid-20th century. IDAD resin emerged as a particularly effective chelating resin due to its unique chemical structure and ability to form stable complexes with a wide range of metal ions.
Today, IDAD resin is synthesized through various methods, including polymerization of iminodiacetic acid with crosslinking agents like divinylbenzene. The resulting polymer structure provides a robust matrix with iminodiacetic acid functional groups strategically positioned for efficient metal ion complexation. Continued research focuses on optimizing synthesis methods to improve resin properties like porosity, selectivity, and mechanical strength.
The global need for effective metal separation and purification is immense, impacting industries across the board. From recovering precious metals in electronics recycling to removing toxic heavy metals from industrial wastewater, the applications are diverse and crucial. Furthermore, the demand for high-purity metals in the semiconductor industry and the increasing focus on circular economy principles are driving further innovation in resin technology, placing iminodiacetic acid resin at the forefront of sustainable solutions.
Geographically, the Asia-Pacific region dominates the ion exchange resin market due to rapid industrialization and stringent environmental regulations in countries like China and India. North America and Europe are also significant consumers, driven by established industries and increasing environmental awareness. The United States Environmental Protection Agency (EPA) and similar regulatory bodies worldwide are continually raising standards for effluent discharge, boosting the demand for IDAD resin-based water treatment systems.
Beyond industrial applications, IDAD resin is also finding increasing use in environmental remediation projects, such as the cleanup of contaminated sites and the removal of radionuclides from nuclear waste. Its versatility and effectiveness make it a valuable tool in addressing a wide range of environmental challenges globally.
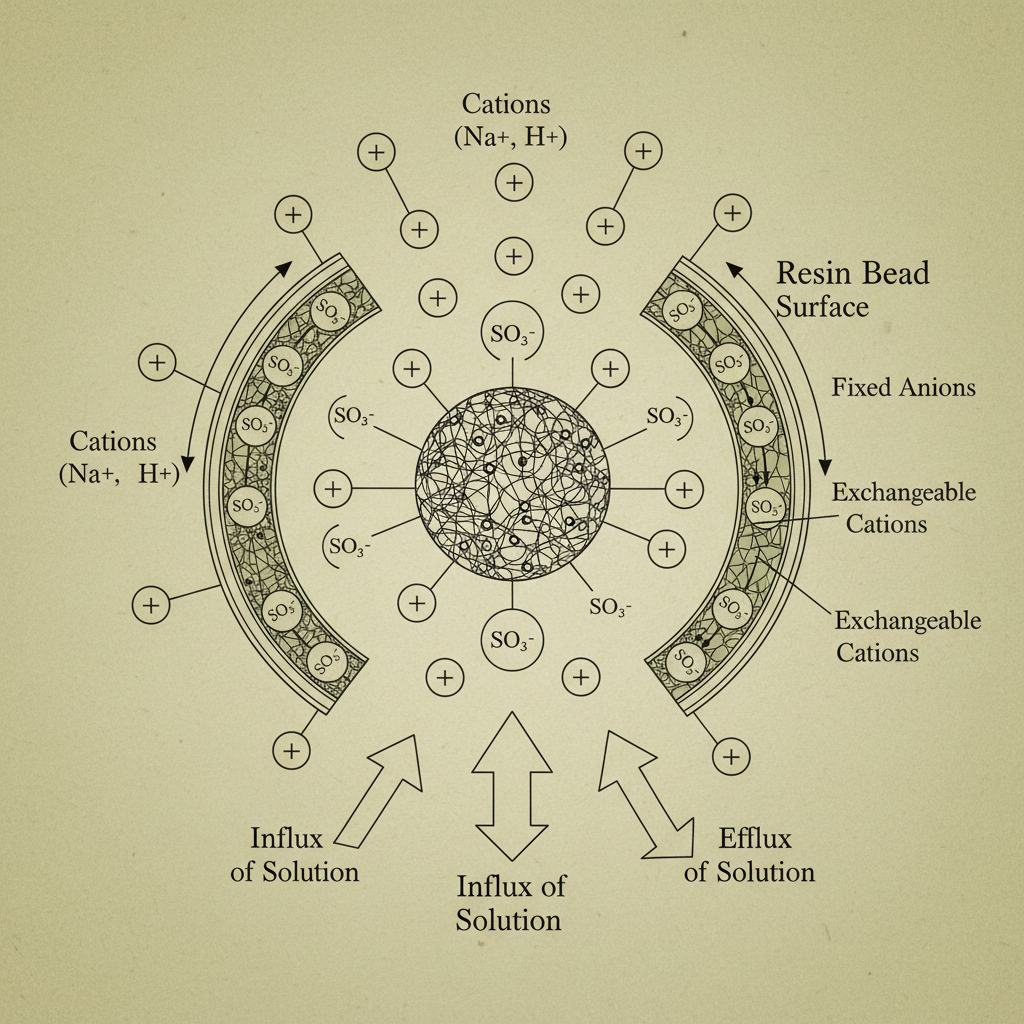
Iminodiacetic acid resin is a type of chelating resin specifically designed to selectively bind metal ions. It's typically a polymeric matrix, usually polystyrene-divinylbenzene copolymer, to which iminodiacetic acid functional groups are chemically attached. These functional groups contain nitrogen and carboxylate moieties, creating a strong affinity for various metal ions through a process called chelation – where the resin forms stable, ring-like complexes with the metal.
Essentially, the resin acts like a molecular “sponge” that selectively absorbs specific metal ions from a solution. The selectivity is determined by factors such as the pH of the solution, the concentration of competing ions, and the specific type of metal ion being targeted. This selective binding capability distinguishes IDAD resin from traditional ion exchange resins, which primarily rely on electrostatic attraction.
Its connection to modern industry and humanitarian needs is significant. In the pharmaceutical industry, it’s used to purify drugs and remove metal contaminants. In environmental science, it’s instrumental in remediating contaminated water sources. Even in the mining industry, it is utilized to recover valuable metals from ore leachates, contributing to more efficient and sustainable resource extraction.
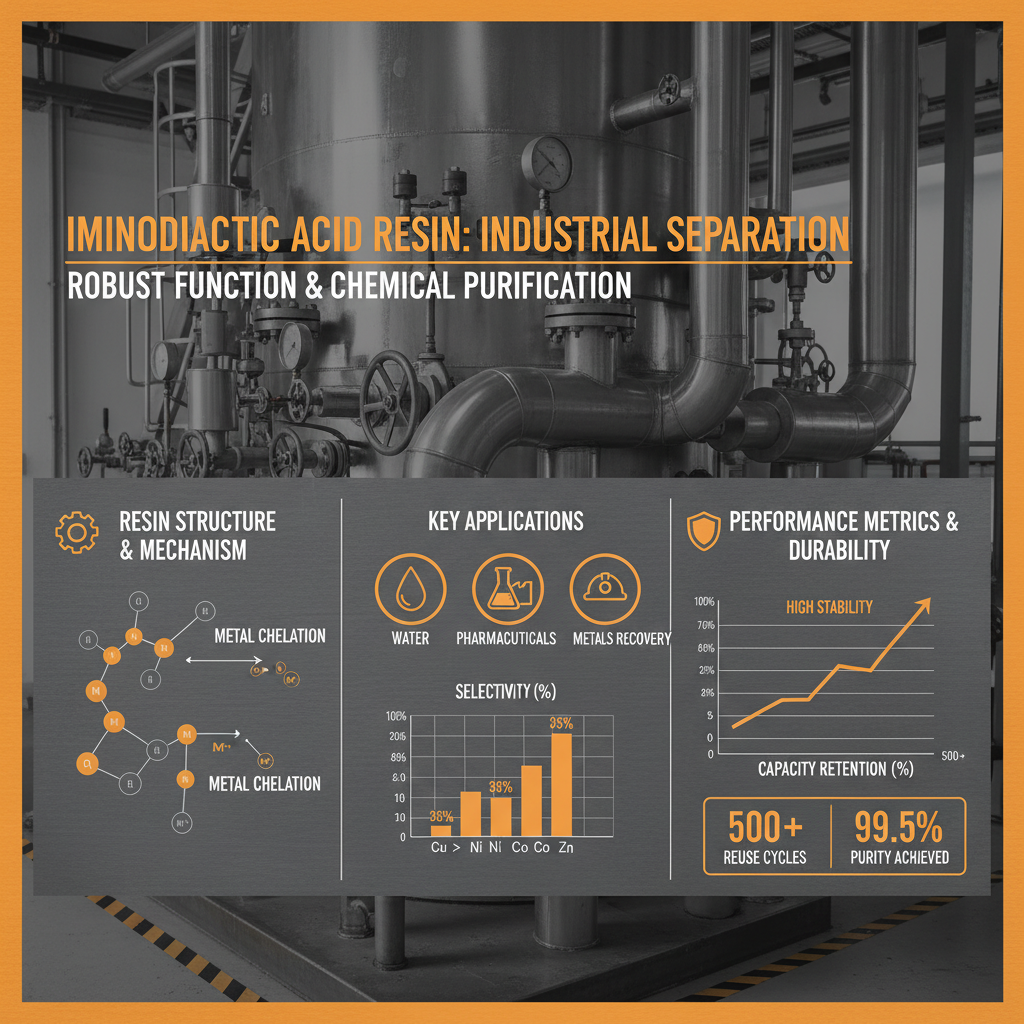
One of the key features of iminodiacetic acid resin is its Selectivity. Unlike standard ion exchange resins, IDAD exhibits a strong preference for certain metal ions over others, allowing for targeted separation and purification. This selectivity is governed by the stability constants of the metal-resin complexes.
Another crucial aspect is its Capacity. This refers to the amount of metal ions the resin can bind per unit volume. Higher capacity resins are generally more efficient and require less material for a given application. Capacity is influenced by the density of iminodiacetic acid functional groups on the resin matrix and the porosity of the resin.
In the mining industry, IDAD resin is employed for the selective recovery of valuable metals such as gold, silver, and platinum from leach solutions. This process reduces environmental impact compared to traditional methods like cyanide leaching. In post-disaster relief operations, IDAD resin can be used in mobile water purification units to remove toxic contaminants from water sources, providing safe drinking water to affected populations.
The pharmaceutical sector leverages IDAD resin for the purification of drug intermediates and the removal of heavy metal impurities from final products, ensuring drug safety and efficacy. Similarly, in the food and beverage industry, it’s used to demineralize fruit juices and remove unwanted metal ions, improving product quality and stability.
The long-term value of using iminodiacetic acid resin stems from its cost-effectiveness, particularly when considering the potential for metal recovery and reuse. By selectively extracting valuable metals, the resin reduces waste and contributes to a circular economy. Its durability and reusability also contribute to lower operational costs over time.
Furthermore, IDAD resin offers significant environmental benefits. By removing harmful pollutants from water and industrial effluents, it protects ecosystems and human health. The reduction in reliance on harsh chemical treatments and the ability to recover valuable resources promote sustainability and responsible resource management.
Future trends point towards the development of "smart" IDAD resins with enhanced selectivity and responsiveness. Nanomaterials are being incorporated into the resin matrix to increase surface area and improve metal ion binding capacity. Moreover, research is focused on creating biodegradable resins to minimize environmental impact at the end of their lifecycle.
Digitalization and automation are also playing a role, with the development of sensor-equipped resin systems that can monitor metal ion concentrations in real-time and optimize separation processes. These advancements, coupled with the increasing adoption of green chemistry principles, will further solidify IDAD resin’s position as a cornerstone of sustainable chemical engineering.
The integration of IDAD resin with membrane technologies is another promising area of innovation, potentially leading to hybrid systems with even greater efficiency and selectivity in metal separation and recovery.
While both types of resins facilitate ion exchange, IDAD resin is a chelating resin, meaning it forms more stable complexes with metal ions due to the presence of iminodiacetic acid functional groups. Traditional resins rely primarily on electrostatic attraction, which is less selective and can be easily disrupted by competing ions. This enhanced selectivity makes IDAD resin ideal for specific metal recovery and purification applications.
The pH of the solution is a critical factor influencing the performance of IDAD resin. Metal ion binding is typically optimal within a specific pH range, dependent on the metal being targeted. At lower pH values, protonation of the iminodiacetic acid groups can reduce their binding capacity. Conversely, at higher pH values, metal ions may precipitate as hydroxides, reducing their availability for chelation. Careful pH control is essential for maximizing resin efficiency.
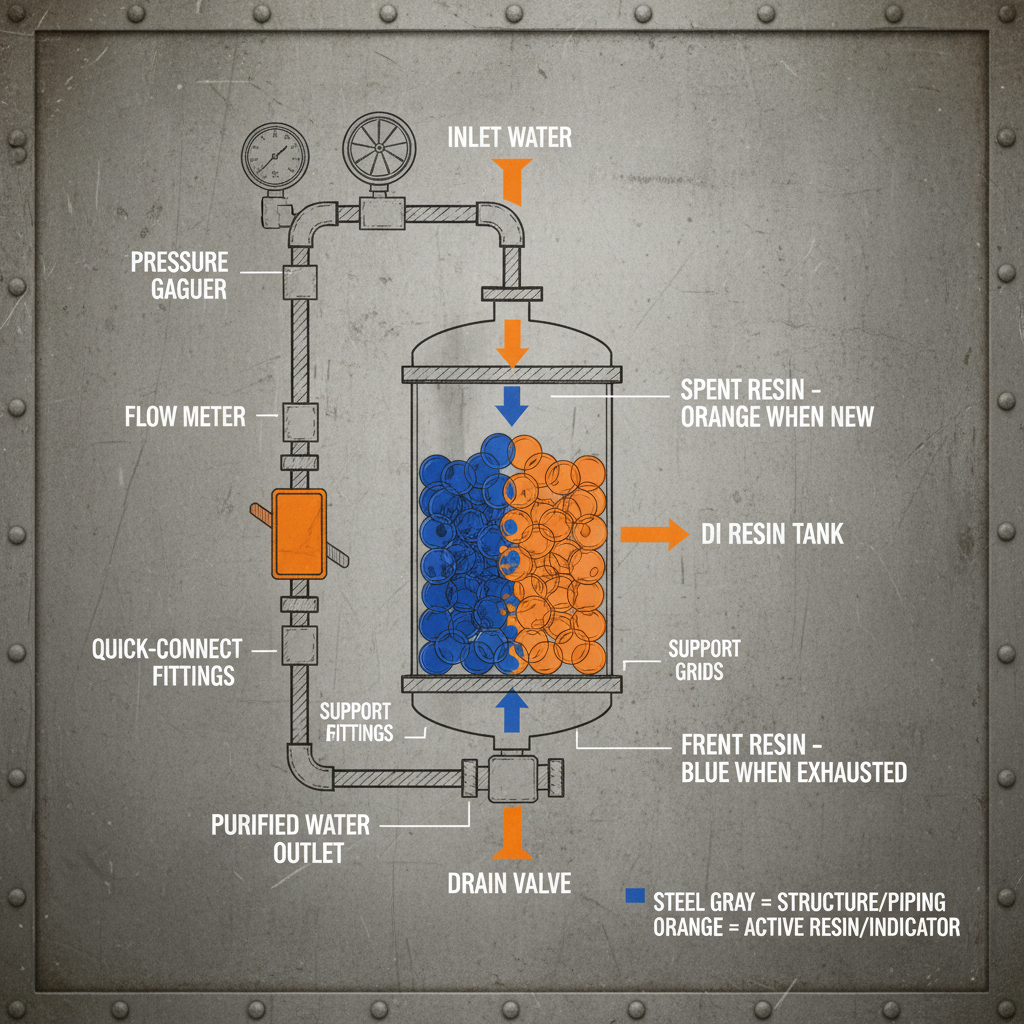
The lifespan of IDAD resin varies depending on operating conditions and the type of metal ions being bound. Generally, with proper maintenance, a resin can last several years. Regeneration involves removing the bound metal ions using a suitable eluent, typically an acidic or complexing solution. Repeated regeneration cycles can gradually degrade the resin matrix, reducing its capacity and selectivity over time.
Compared to some traditional metal separation techniques, IDAD resin offers a more environmentally friendly solution. It reduces the use of hazardous chemicals and facilitates metal recovery and reuse, minimizing waste. However, the resin itself is typically a synthetic polymer, so proper disposal or recycling is crucial to avoid environmental contamination. Research into biodegradable resin alternatives is ongoing.
Some common challenges include fouling of the resin surface by organic matter or suspended solids, which reduces its capacity and selectivity. Competition from other ions in the solution can also hinder the binding of target metal ions. Furthermore, the cost of regeneration eluents and the potential for resin degradation over time can contribute to operational expenses. Effective pretreatment of the feed solution and careful optimization of operating parameters are crucial for mitigating these challenges.
We at Liji Resin specialize in the production and supply of high-quality iminodiacetic acid resin for various applications. We offer a range of resin types with different properties to meet specific customer needs. Contact us today for a quote or to discuss your requirements.
Iminodiacetic acid resin represents a powerful and versatile tool for metal separation, purification, and recovery. Its unique chelating properties, combined with its durability and reusability, make it an essential component in a wide range of industries, from mining and pharmaceuticals to environmental remediation. The ongoing advancements in resin technology, driven by sustainability concerns and increasing demand for resource efficiency, promise even greater performance and applications in the future.
Looking ahead, continued research and development are crucial to unlock the full potential of IDAD resin. The development of biodegradable resins, the integration of nanomaterials, and the implementation of smart monitoring systems will further enhance its effectiveness and environmental compatibility. To learn more about how iminodiacetic acid resin can benefit your operations, visit our website today.