







Honestly, things have been moving fast in the construction materials world lately. Everyone's chasing "smart" and "green," which is good, but it also means a lot of hype. I spend most of my year bouncing between factories and job sites, and you start to see patterns. There’s a real push for prefabrication, more modular stuff... less cutting and welding on-site, which the guys appreciate. But that also means tighter tolerances, more reliance on precise manufacturing.
Have you noticed everyone talking about composite materials? It's all the rage. And that’s fine, but a lot of times, these “innovations” end up being a pain to actually work with. I encountered this at a precast concrete factory last time. They switched to a new polymer additive to supposedly increase strength, and the guys were complaining it made the mix so sticky, it gummed up all the machinery. It's not enough to just have good numbers in a lab report.

To be honest, the demand for ion exchange resin has been steadily climbing for years, but it’s really kicked into high gear recently. It’s not just water purification anymore; industries like pharmaceuticals, food processing, and even power generation are relying on it more and more. The UN estimates that global water stress will intensify significantly in the next decade, and that’s a massive driver for technologies like ion exchange.
It’s funny, a lot of people think of it as some complex chemistry thing, but at its core, it’s just about cleaning up stuff. And everyone needs clean stuff. We’ve seen increased orders from regions facing severe water scarcity – parts of Africa, the Middle East, even the American Southwest. There's a real need to reclaim and reuse water, and ion exchange resin is a key part of that.
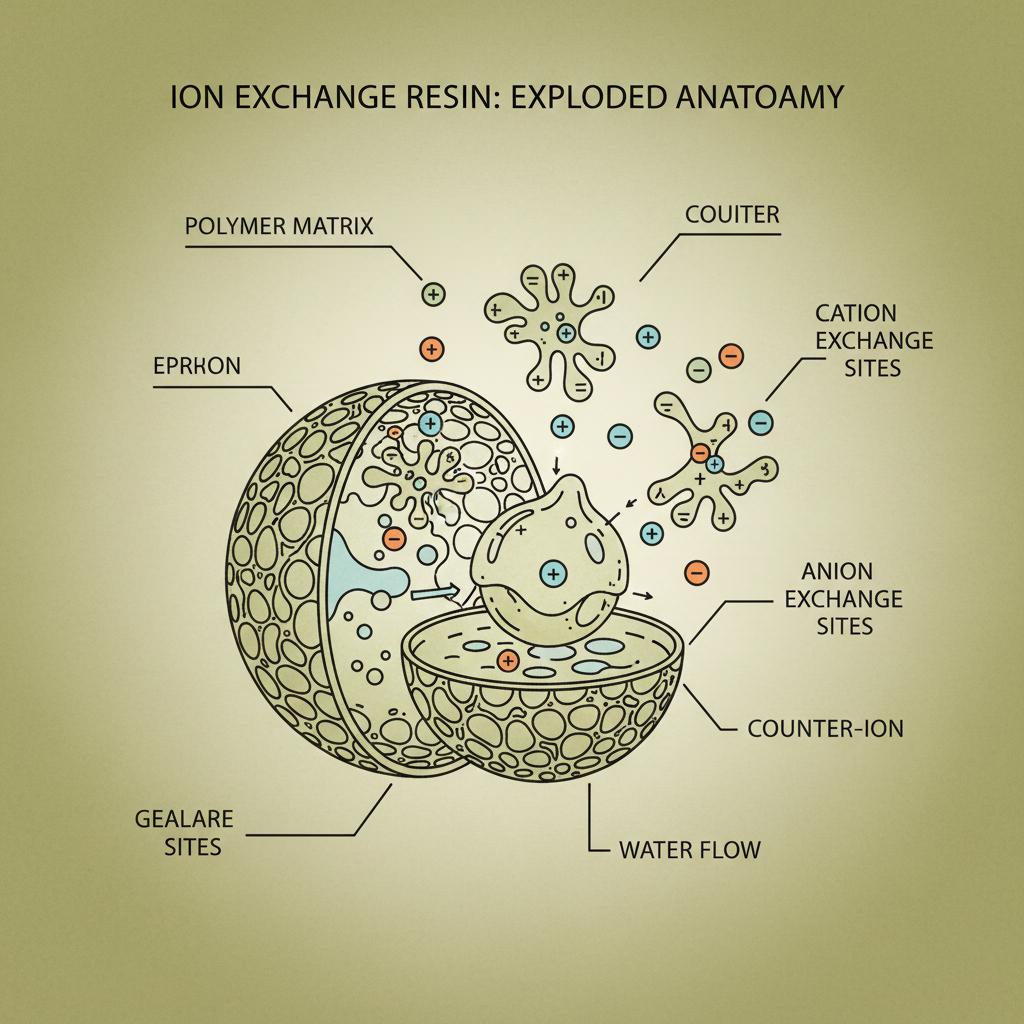
So, what is ion exchange resin? Simply put, it’s a material – usually a tiny plastic bead – that can selectively remove unwanted ions from a liquid. Think of it like a microscopic filter, but instead of physically blocking particles, it swaps ions. It's how you get soft water, remove nitrates from drinking water, and purify chemicals.
It connects to modern industry in pretty much every way imaginable. Power plants use it to prevent corrosion, pharmaceutical companies use it to purify drugs, and food processors use it to remove impurities from sugar. It's even crucial in nuclear waste treatment, removing radioactive elements. Strangely, a lot of folks outside of engineering don’t even realize how widespread its use is.
It’s critical for humanitarian efforts too. Providing access to clean drinking water in disaster relief situations or developing countries often relies on portable ion exchange systems. It’s a reliable and relatively cost-effective way to make water safe to drink when other options aren’t available.
There are a few key things that dictate how well an ion exchange resin performs. First, there's the matrix – the physical structure of the bead. It needs to be strong enough to withstand the pressure and flow rates of the system. We use polystyrene-divinylbenzene copolymers primarily. They smell faintly of plastic – a little chemical, but not awful. It's that smell that tells you it's good quality.
Then there’s the functional group – the part of the bead that actually grabs the ions. This is where the chemistry gets interesting. Different functional groups target different ions. Sulfonic acid groups are great for softening water, while amine groups are used for removing heavy metals. We handle these materials carefully, following strict safety protocols, because some of those reagents can be pretty nasty.
Capacity is another big one – how much resin you need for a given amount of contamination. And then there’s selectivity – how well it targets the specific ions you want to remove. It’s a balancing act. A high-capacity resin might not be very selective, and vice versa. Durability is also key; you want a resin that can withstand repeated cycles of regeneration without falling apart.
Honestly, the applications are almost endless. In power generation, they prevent scale buildup in boilers. In the food industry, they demineralize fruit juices. In the semiconductor industry, they purify water used in manufacturing microchips.
I was at a lithium mining operation in Chile last year, and they were using ion exchange to separate lithium from other elements in the brine. It’s a critical part of the battery production process. And in remote industrial zones where access to clean water is limited, portable ion exchange units are a lifesaver.
Even in post-disaster relief operations, these resins are used in field-deployable water purification systems. It’s a versatile technology that adapts to a wide range of needs.
The real benefit of ion exchange resin isn’t just about getting clean water or purified chemicals; it’s about long-term cost savings and sustainability. Compared to other purification methods, it can be surprisingly economical, especially when you factor in the ability to regenerate the resin and reuse it.
It’s also a relatively energy-efficient process. And with growing concerns about environmental impact, being able to reduce waste and conserve resources is a big win. The reliability is also a huge factor. Properly maintained systems can operate for years with minimal downtime.
Anyway, I think the biggest trend right now is developing more selective resins. We're seeing a lot of research into materials that can target specific contaminants, like PFAS ("forever chemicals"), with greater efficiency. There’s also work being done on bio-based resins – using renewable resources instead of petroleum-based materials.
The integration of ion exchange with other technologies, like membrane filtration and advanced oxidation processes, is also gaining traction. And the use of AI and machine learning to optimize resin regeneration and predict performance is starting to emerge. It's interesting stuff.
To be honest, it's not all smooth sailing. Fouling is a constant headache. Organic matter and other contaminants can coat the resin beads and reduce their efficiency. Regular cleaning and pretreatment are essential, but it’s an ongoing battle.
Regeneration can also be tricky. You need to use the right chemicals in the right concentrations, and you have to deal with the resulting waste stream. We’re working on developing more sustainable regeneration methods, using less harsh chemicals. And another challenge is the disposal of spent resin. Finding environmentally responsible ways to recycle or dispose of it is a growing concern.
Ultimately, it all comes down to understanding the specific application and tailoring the system to meet the needs. There’s no one-size-fits-all solution.
| Resin Type | Flow Rate (m/hr) | Regeneration Frequency | Operating Cost (USD/m3) |
|---|---|---|---|
| Strong Acid Cation | 10-20 | Daily/Weekly | 0.50-1.00 |
| Strong Base Anion | 15-25 | Bi-Weekly/Monthly | 0.75-1.50 |
| Weak Acid Cation | 20-30 | Monthly/Quarterly | 0.30-0.80 |
| Chelating Resin | 5-10 | As Needed (Metal Load) | 1.00-2.50 |
| Mixed Bed Resin | 15-25 | Weekly/Bi-Weekly | 0.60-1.20 |
| Selective Resin (PFAS) | 8-15 | Monthly/Quarterly | 1.50-3.00 |
The lifespan really depends on the application and how well the system is maintained. Under ideal conditions – consistent water quality, proper regeneration – a good quality resin can last 5-10 years. But things like excessive fouling, mechanical stress, or chemical attack can significantly shorten that lifespan. We've seen resins fail in as little as a year in heavily contaminated environments, while others have lasted 15 years with careful management.
There are several telltale signs. A drop in effluent quality is a big one, even after regeneration. You might also notice increased pressure drop across the resin bed, indicating fouling. And if you're seeing a significant amount of resin attrition – basically, the beads are breaking down – that's a clear sign it's time for a replacement. Regular resin analysis can also help you monitor its condition and predict when replacement is needed.
It's a mixed bag. The resin itself is a plastic, so there's a sustainability issue there. But compared to other water treatment technologies, it can be a more environmentally friendly option. It doesn't require high energy consumption, and it can significantly reduce the discharge of pollutants. The biggest environmental concern is the disposal of spent resin and the chemicals used for regeneration. That’s why we are pushing to develop more sustainable regeneration techniques and investigate resin recycling options.
Type 1 water is ultra-pure water used in very sensitive analytical applications, like pharmaceutical research or semiconductor manufacturing. Type 2 water is high-purity water used for things like rinsing lab equipment. Ion exchange is critical for achieving both. For Type 1, it’s used in combination with other technologies like reverse osmosis and UV sterilization to remove almost all contaminants. For Type 2, it's often the primary purification method. The difference lies in the degree of purification and the number of stages involved.
Absolutely. That’s actually one of its biggest strengths. We worked with a beverage company last year who needed to remove a very specific organic compound from their fruit juice. They wanted a resin with a much higher selectivity than what was commercially available. We were able to modify the functional group on the resin to target that specific molecule, and it solved their problem beautifully. Customization can involve altering the resin matrix, the functional group, or the bead size.
Fouling is a major one, as mentioned before. Also, it’s not effective at removing all types of contaminants. Things like bacteria and viruses require additional treatment. And while it's relatively energy-efficient, regeneration does require chemicals, which can add to the cost and environmental impact. We’re constantly working to address these limitations, but it’s important to be realistic about what ion exchange can and can’t do.
Ultimately, ion exchange resin is a powerful and versatile technology with applications spanning a huge range of industries. It’s not a silver bullet, and it has its limitations, but when applied correctly, it can deliver significant cost savings, improve sustainability, and enhance product quality. We’ve come a long way from the early days of simple water softening, and the future looks bright with ongoing innovations in materials and applications.
But here’s the thing: all the chemistry and engineering in the world don’t matter if the system isn’t properly installed and maintained. It’s the worker on the ground, tightening the last screw, monitoring the flow rates, and adjusting the regeneration cycles, who ultimately determines whether this thing works or not. That’s the bottom line.





